Point-of-Care (POC) Diagnostic Testing: 5 Key Trends to Watch from 2022-2026
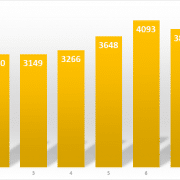
Globally, millions of dollars are spent annually on point-of-care (POC) diagnostic testing, both professional testing (which includes physician office labs, clinic labs, employer labs and hospital labs) and self-testing (which includes home, drug store testing and continuous glucose testing). In 2021, amidst the pandemic, the POC market reached $41,149 million ($41.1 billion) and the market is forecast to grow through 2026, according to The Worldwide Market for Point-of-Care (POC) Diagnostic Tests, 9th Edition, a new report by leading medical market research firm Kalorama Information.
COVID-19 was a significant game changer in 2020-2021. The number of new tests for POC COVID-19 was stunning. In addition, new technologies and increased demand contributed to growth in the markets. Previously, in some segments, COVID-19 caused a contraction due to lockdown, closures, limited physician visits and a reduction in elective and outpatient surgery. However, the market has rebounded and is expected to resume pre-pandemic growth in all segments of the POC market.
Diagnostic tests performed outside the central laboratory or decentralized testing is generally known as POC, including rapid testing. POC tests are performed onsite in a medical facility while rapid testing can be run in various types of medical settings or at home. Over the years, the increasing introduction of transportable, portable, and handheld instruments has resulted in the migration of POC testing from the hospital environment to a range of medical environments including the workplace, home, disaster care and most recently, convenience clinic. Other terms used to describe POC testing include near-patient testing, physician’s office laboratory (POL) testing, rapid testing, patient self-testing or patient self-management, bedside testing, remote testing, or satellite testing.
One of the key benefits and market drivers for the POC testing market is the ability to have rapid results that can be used immediately for patient treatment decisions. While this is a major benefit of POC testing, many other factors are also driving growth of this market. Other factors and trends driving the development of POC tests include, but are not limited to, the following:
1. Patient focused drivers and trends
- Demand for faster turnaround times for critical test results in hospital locations such as emergency departments and intensive care units.
- Less sample volume (neonatal, pediatric, ICU benefit).
2. Medical/Healthcare related drivers and trends
- Introduction of POC tests for new targets such as additional infectious diseases and other new analytes, which previously could not be tested in the POC setting.
- Growth of personalized healthcare and personalized medicine, and the need for rapid tests to support this.
3. Technology drivers and trends
- Advances in miniaturization, nanotechnology, microfluidics, molecular and other technologies that have made it possible to expand the numbers and types of tests that can be performed in POC settings.
- Health apps for mobile devices are pushing the development of POC devices.
4. Economic drivers and trends
- Growth in specialty clinics, retail clinics, and other decentralized locations for patient care.
- Economic factors driving the need to reduce the length of patient stays in hospitals, reduce backlog in areas such as emergency departments as patients wait for test results, etc.
5. Other drivers and trends
- Need for robust but also simple diagnostic tools for military applications outside of military hospitals.
- Need for disaster preparedness.
- Need for robust but also simple diagnostic tools for developing countries.
- Need for pandemic preparedness
For more information or to purchase The Worldwide Market for Point-of-Care (POC) Diagnostic Tests, 9th Edition, visit: https://kaloramainformation.com/product/the-worldwide-market-for-point-of-care-poc-diagnostic-tests-9th-edition/.